Author Archive
Hello, hello. You’re now tuned to your favorite blog: micro-writers.egybio.net. Tonight we have this very special guest, live, online. After two months of waiting, we finally got this exclusive interview with the emerged Streptococcus pyogenes strain, the most dangerous ever, M1T1. We have it here, with us, in the studio.
– Hello, M1T1. Welcome in our studio.
– Hey there.
– We knew from our resources, which are totally classified, that you got yourself in trouble recently.
– (Interrupting), I did NOT get myself in trouble. EID set me up.
– M1T1, Would you please calm down & tell us a little more about yourself?
– Well, I belong to Group A streptococci (GAS) aka Streptococcus pyogenes. M1T1 is my serotype; I’m just a clonal strain. As you know, S. pyogenes colonize human skin & throat causing either non-invasive (sore throat, tonsillitis & impetigo) or invasive (necrotizing fasciitis NF, scarlet fever & streptococcal toxic-shock syndrome STSS) infections. Actually, NF gave me my nick: Flesh-eating bacteria.
– So, you cause all people NF & STSS?
– No, kid. It depends on their genetic susceptibility, what you call “Host–pathogen interactions”. I was isolated from patients with invasive as well as non-invasive infections during 1992–2002. This is NOT entirely my fault; humans can make me extra virulent by selecting the most virulent members.
– Back to your history, when have you exactly been isolated?
– M1 & her sisters were the worst nightmare in US & UK in the 19th century as they caused the famous pandemic of scarlet fever. “Nevertheless”, early 1980s was the golden age of my strain as well as my very close sisters M3T3 & M18. We caused STSS & NF in different parts of the world. Great times, great times!
– Only for you, I suppose! So, what made you hypervirulent? What caused you this “epidemiologic shift”?
– Two reasons Dr Ramy K. Aziz identified that improved my fitness to humans: the new genes I got from phages & “host-imposed pressure”. Both resulted in the selection & survival of me M1T1 the hypervirulent strain. Dr Aziz’s work at Dr Kotb’s lab resulted in identification of a group of genes I got from phages that changed my entire life.
– Interesting! Tell us more about that. How did phages “change your life”?
– Dr Aziz proved that I differ from my ancestral M1 when he found that I have 2 extra prophages (lysogenized phages didn’t get the chance to lyse me, so they became integrated in my genome):
1. SPhinX which carries a gene encodes the potent superantigen SpeA or pyrogenic exotoxin A (scarlet fever toxin).
2. PhiRamid which carries another gene encodes the most potent streptococcal nuclease ever, Sda1.
3. He also found that phages conversion from the lytic state to the lysogenic state resulted in exchange of toxins between our different strains (aka Horizontal Gene Transfer). Phages are very good genetic material transporters, what makes “strains belonging to the same serotype may have different virulence components carried by the same or highly similar phages & those belonging to different serotypes may have identical phage-encoded toxins.” What a quote from Rise and Persistence of Global M1T1 Clone of Streptococcus pyogenes.
– Well, It was not that interesting. So, what? What’s the significance? How that made you hypervirulent?
– You can’t get it? You’re not that smart, are you? Tell me, what made M1 hypervirulent causing scarlet fever in the 1920s and me hypervirulent causing STSS in the 1980s with a 50-years decline period?
– Superantigen?
– Exactly. You do have your moments! Superantigen encoding-gene was present in us and absent in strains isolated in the period between them. The interesting part, for me of course, that humans after 50 years of absence of hypervirulent strains had absolutely no superantigen-neutralizing antibodies. That was the real invasive party. Superantigen causes high inflammatory response because of its non-specific binding to immune system components (antibodies & complements) causing an extremely high inflammatory response. In fact, SuperAg inflammatory response is “host-controlled”.
– So, what about Sda1?
– Streptodornase (streptococcal extracellular nuclease) helps me to degrade neutrophils that entrap me in the neutrophil extracellular traps (NETs). So, I can invade humans freely & efficiently and be able to live in their neutrophils. Dr Aziz proved in his paper “Post-proteomic identification of a novel phage-encoded streptodornase, Sda1, in invasive M1T1 Streptococcus pyogenes” that it’s all about C-terminus in my Sda1; the frame-shift mutation increased my virulence while deletion decreased it.
– Now we know about your SuperAg & nuclease (DNase), what’s the “host-imposed pressure”?
– I have my own SpeB (Protease), I use it to degrade my other proteins (virulence factors), which provides me with a good camouflage & gives me access to blood. When the host immune system recognizes me, it traps me in NETs. At this time, I secret Sda1 to degrade neutrophils. Actually, SpeB protects you, humans, from my Sda1& my other toxins. When SpeB was compared in patients with severe & non-severe strep infections, it was found that SpeB wasn’t expressed in case of severe infection. Expression of SpeB may be host-controlled, as host selects the mutants with a mutation in covS, a part of my regulatory system which regulates my gene expression including SpeB gene.
– Finally, M1T1. How do you see your future?
– More new phage-encoded genes, more selection of the hypervirulent strains by the host & more regulation of expression of my virulence factors. Pretty good future! I also count on humans to not develop immunity against me like what happened in 1980, when I got new virulence factors or allelic variations in my old ones.
Thank you, M1T1. Pleasure talking to you…….M1T1? M1T1, where are you? Why do I feel this strange pain in my throat?
Image credits:
Streptococcus pyogenes: http://adoptamicrobe.blogspot.com/
Tags: covS, epidemiologic shift, flesh-eating bacteria, global M1T1 clone, group A streptococci (GAS), horizontal gene transfer, host-imposed pressure, host–pathogen interactions, impetigo, M1T1, Malak Kotb, necrotizing fasciitis, neutrophil extracellular traps (NETs), phage-encoded toxins, pyrogenic exotoxin A, Ramy K. Aziz, S. pyogenes, scarlet fever, sore throat, streptococcal nuclease Sda1, streptodornase, STSS, superantigen SpeA, tonsillitis
 5 Comments »
5 Comments »
It’s hard to talk about someone you know who passed away, but, believe me; it’s harder when you talk about someone you know nothing about. To know someone better, you have to read his writings or, simply, read what his friends wrote about him. Reading what a disciple of his, David M. Morens the director of the National Institute of Allergy and Infectious Diseases, wrote about him made me believe that Dr. Gregg is the luckiest person on the planet by gaining this undeniable love & appreciation from his students. Dr. Gregg died on July 9, 2008 at the age 78.
Michael B. Gregg, M.D. was born in 1930 in Paris, France. He was educated at Stanford University and Western Reserve University School of Medicine. He entered the Public Health Service in 1959 right after he completed his residency in internal medicine at Columbia Presbyterian Hospital in NY. He first served at the National Institutes of Health Rocky Mountain Laboratory. He then trained in infectious diseases in Lahore, Pakistan before he joined CDC (it was known as the Communicable Disease Center) in 1966 as Chief Epidemic Intelligence Service Officer (EISO). From 1967-1988, Dr. Gregg (or Mike like everyone in the CDC used to call him) was the editor of the MMWR. His writing style “just the facts” made thousands of epidemiologists believe that good medical writing indicates clear thinking, which is the only thing needed in Epidemiology. In 1975, Mike was the MMWR editor, Viral Diseases Division director and deputy director of the Bureau of Epidemiology.
Mike was not only a MMWR editor, but he taught hundreds of students in NIH & CDC to become leaders in epidemiology & public health. Mike’s textbook “Field Epidemiology” (Don’t get extremely excited, It’s a link to the review not the book itself. My apologies.) is the “go to” book in breakout investigation & to solve public health issues. As David M. Morens says, it reflects his very wide experience in national as well as international epidemiology including:
- Pontiac fever/Legionnaires’ disease (1968/1976) [Ligeonella pneumophila – Philadelphia] First, it was thought to be a 1918-like influenza pandemic, but Mike kept running through the history of Influenza & other respiratory diseases, the epidemic ones. After a couple of days of collecting info, Mike was the first to say: “This is beginning to look like Pontiac fever.”
- Swine flu (1975–1976) [Hsw1N1 – New Jersey]
- Guillain-Barré syndrome (1977)
- Ebola hemorrhagic fever (1976) [(-)ssRNA virus – Zaire & Sudan]
- In the June 5, 1981 issue, he published a report about 5 Pneumocystis carinii pneumonia cases. Such disease was rare, so he wrote a note saying: “the case histories suggested a cellular-immune dysfunction related to a common exposure, a disease acquired through sexual contact”, which we know now as HIV/ AIDS. It was the first report about it.
- He helped in putting on the epidemiology map of Reye syndrome (1973–1977) [A fatal disease associated with Aspirin consumption when having a viral-disease e.g. Varicella – Ohio] , Kawasaki disease (1977), and toxic-shock syndrome (1980) [S. aureus]. He was consulted in the SARS outbreak in 2003 [Severe acute respiratory syndrome (+)ssRNA coronavirus] even after his full retirement.
Mike used to give the epidemiology course each July. The first subject was: “How to investigate an epidemic”. His first words were: “First, you need to find a good map…” He was teaching his students to keep an open and interested mind, to remain flexible and creative, to rethink and to assemble the puzzle pieces quickly to get the big picture.
“It’s better to be approximately right today than exactly right tomorrow,” a phrase said by Michael O’Leary, a former epidemiology student of Mike rephrasing his description of epidemiologists’ work: “Quick and dirty”.
Image credits:
Michael B. Gregg: http://www.cdc.gov/
Tags: ebola hemorrhagic fever, field epidemiology, Guillain-Barré syndrome, Michael B. Gregg, MMWR, Pneumocystis carinii, pontiac fever, reye syndrome, SARS outbreak, swine flu, toxic-shock syndrome
 2 Comments »
2 Comments »
Who could possibly believe that our anaerobic spore-forming Clostridia will be used as an anti-tumor therapy?! It’s called Clostridium-based tumor targeted therapy. “Give me a break,” that’s exactly what I said when I read the review of the new book Clostridia: Molecular Biology in the Post-genomic Era.
So, how does it work? There are various non-pathogenic Clostridia strains which could replicate within solid tumors upon systemic administration. The interesting part is coming right up: Why solid tumors?! It’s because of its very unique physiology; it characterized by hypoxia & necrosis which totally fits the anaerobic Clostridia. The advantage will be the selectivity & targeting of the cancer cells leading to destroying them.
It was news to me to know that Cl. perfringens causes food poisoning like any food-borne illness & causes antibiotic-associated diarrhea (When I hear the name Cl. perfringens, I orient myself toward gas gangrene right away). Its enterotoxin gene (cpe) is present on the chromosome itself (in food poisoning isolates) and on the plasmid (in the antibiotic-associated diarrhea isolates). The enterotoxin binds to claudin receptors, then there’s oligomerization or “prepore” formation & finally prepore insertion takes place to form the functional pore which kills the cells by apoptosis. So CPE/CPE derivatives could be used for cancer therapy.
Tags: anti-tumor, clostridia, Clostridium-based tumor targeted therapy, CPE/CPE, enterotoxin, solid tumors
 2 Comments »
2 Comments »
Sept 16th:
Hi, Diary;
Now it’s in the press. It’s humiliating. In Scientific American, it was written under the title: Turning Bacteria into Plastic Factories. So that’s what we, E. coli, turned into: slaves for humans to do whatever they want us to do.
I remember this like it happened yesterday. It was someday in the beginning of the year 2008. That day we found ourselves in those tanks, large ones, filled with sugar & water. “This is great; we can ferment the whole amount of sugar in the tank. This is the perfect place to live in,” that is what we thought. Then we got this strange order from our genomes or plasmids, I’m not sure & found ourselves producing that enormous amount of 1, 4- Butanediol (BDO). This was so strange because BDO used to be toxic to us at low levels (I’ve heard once that any production of a non-native material inhibits our growth). Then we realized the truth; we’ve been genetically engineered to tolerate BDO, the raw material for a very large number of plastic, rubber and fiber products including solvents, fine chemicals, pharmaceuticals, automotive components, electrical and electronics components, as well as apparel fibers.
Honestly, I don’t know. Sometimes I feel that bioengineers from San Diego-based Genomatica, Inc. are right after all; they modified us, they wanted to make use of us, they wanted to save money & energy needed to produce BDO from non-renewable petrochemical feedstocks, the currently used method. Plus it causes us no harm as Bioengineer Christophe Schilling, president and co-founder of the company said: “We have engineered the organism such that it has to secrete that product in order for it to grow.”
Now I know what it feels like with my fellow bacterial species, the natural born producers. Most bacteria synthesize the organic polyesters Polyhydroxyalkanoates (PHAs) to be used as a carbon & energy storage material. Now they’re discussing the ability to use these PHAs as biodegradable plastics.
 Source: A piece of a plastic notebook found in the Petri dish appears in the picture below, Genomatica labs, San Diego. Source: A piece of a plastic notebook found in the Petri dish appears in the picture below, Genomatica labs, San Diego.
Image credits:
Genetically engineered E. coli may produce plastics: http://www.sciam.com/
Tags: BDO, biodegradable plastics, Christophe Schilling, genetically engineered, Genomatica, PHAs
 3 Comments »
3 Comments »
“It’s an honor to announce that we’ve successfully discovered, cloned & characterized the densonucleosis virus (DNV) Antigen, which will be able to infect Anopheles gambiae. That will hinder its ability to transmit Malaria parasite, Plasmodium or, at least, reduce its lifespan. I’ve to tell you this: This will be the latest in Malaria control.” Well.. those are my words but, of course, it is not my discovery. It’s the team of researchers from Johns Hopkins who discovered it. To realize how big it’s; we first need to know the previous approaches & trials in Malaria control.
To think, just think, about control/ prevention of any of those vector-borne parasites; automatically researchers think about these:
1- Prevent infection – disease – transmission.
2- By looking at: the parasite’s life cycle – the mosquito – the human immune system.
3- So, it’ll look like that: get humans vaccinated – develop genetically modified mosquitoes incapable of transmitting the parasite or simply “I was so naive to think that it’s simple” kill them with insecticides (anti-vector measures) – target the parasite at any stage of its life cycle.
For decades, DDT (dichloro-diphenyl-trichloroethane) & Chloroquine were successfully used in eradication of Anopheles & Plasmodium respectively, “I do like this word, makes me sound like a pro”. Chloroquine was used in treatment as well as prevention, till the emergence of chloroquine-resistant Plasmodium parasites and DDT-resistant Anopheles mosquitoes. Yes, they overcame the humans’ arsenal. So, preventing spread of resistant parasites is the #1 priority.
We can’t talk about all control strategies today, so we’ll talk about anti-vector measures. What do they use in control programs as anti-vector measures?
1- Insecticide-treated bednets (ITNs) & long-lasting ITNs (LLINs) it helped kids to survive. The only allowed insecticides to be used in ITNs are pyrethroid insecticides, so when the pyrethroid resistance emerged, as usual, it was really bad.
2- Indoor residual spraying (IRS) using DDT
One word about the mechanism of action of DDT & pyrethroids, both target voltage-gated Na-channels. So, when a set of mutations change the protein structure; Congratulations! It’s resistance to both DDT & pyrethroids.
3- New approach: Molecular talk; Know more about “blood meal” host selection. Yes, Anopheles smells the host, so researchers want to identify that pathway.
4- Another new approach: They are investigating genes which encode proteins that may interrupt the development of the parasite in the Anopheles.
The latest as an anti-vector measure is using Paratransgenesis or “the genetic manipulation of insect symbiotic (mutualistic, commensal or parasitic) microorganisms”, I can’t get the term or the definition. I’ll say it like that: “Any other m.o. has a relationship with the vector”. The steps are:
1- Know the Ag (Pick the gift)
2- Get the gene(s) engineered to be successfully expressed (Wrap the gift)
3- Delivery to Anopheles (Deliver the gift)
So the gift will be the non-enveloped ssDNA virus called (DNV). Its genome is very small, when they say for a viral genome that it’s small, so it has to be small (4–6 kb). The entire genome can be placed in a plasmid.
Back to the story of the discovery, they were doing a totally unrelated experiment when they found that strange band/ zone. They isolated it from the gel, cloned, sequenced, ran through BLAST which showed that it looks like DNV of Aedes aegypti (AeDNV) but not the man himself. They did multiple tests to identify the Ag, e.g. Immunofluorescence assay. There was a trial to infect Anopheles gambiae with DNV of Aedes aegypti which wasn’t successful in infecting adults from Anopheles gambiae. But the novel “AgDNV is highly infectious to An. gambiae larvae, disseminates to adult tissues, and is passed on to subsequent generations.”
Tags: AeDNV, AgDNV, anopheles gambiae, anti-vector measures, DDT, IRS, ITNs, malaria, paratransgenesis, plasmodium
 No Comments »
No Comments »
You may remember HBV, the famous hepatitis virus with its partially double-stranded circular DNA genome. I always wondered: What is that supposed to mean?! HBV has a very complicated replication cycle. I’m pretty sure that all molecular biology fans will be totally thrilled by reading this.
HBV replication cycle is divided into 3 stages:
1- The infectious virion containing the partially double-stranded circular DNA, they call it RC-DNA (relaxed circular).
2- Right after the infection, inside the host nucleus, the genome becomes cccDNA (covalently closed circular DNA). It looks just like plasmids. HBV needs that highly stable form because it’s a chronic infection; it doesn’t want to be lost during host cell division. It may be still there in the host cells even after effective antiviral therapy.
3- Finally transcription takes place, several RNA molecules are produced, some of them are genomic (contain the whole genome) named pgRNA (pregenomic RNA) & some are subgenomic (encode needed enzymes) It uses the cell’s RNA polymerase II to do all this.

So, what happens to the pgRNA? They get inside progeny capsids ready to be reverse transcribed with the help of P protein (Its reverse transcriptase) which is “co-packed” in the pgRNA- progeny capsid package to get it back to the RC-DNA. Then the mature RC-DNA containing-nucleocapsids could undergo cccDNA amplification, or could be enveloped & ready for release from the cell. Of course all this is in equilibrium; if there’s only one copy in the cell, the priority is not to make cccDNA but to be enveloped & released.
Why the RC-DNA needs to be first cccDNA before transcription? As I got from this review, the RC-DNA has the normal (-)-strand (opposite sense to mRNA) but its complementary, the (+)-DNA strand, is not in full length. It results from the non-identical nucleotides supply; because the envelop is impermeable to nucleotides. At the 5′ end of the (-)-strand, there’s the P protein. But at the 5′ end of the (+)-strand, there’s some RNA nucleotides remains from the pgRNA…It was its primer, remember? All these are removed to be a cccDNA. The P protein may has a role in completing the (+)-strand.
Image credits:
Hepatitis B Virus Replication: http://www.meds.com/
Tags: cccDNA, HBV, P protein, pgRNA, RC-DNA, reverse transcription
 No Comments »
No Comments »
 Two years ago, the project of sequencing the Neanderthal genome started. They (Max Planck Institute & 454 Life sequencing) promised to end by this year. Well, they kept their promise. Frankly, some mitochondrial DNA sequences (mtDNA) have been published but contamination was the major defect in those published sequences. They collected more than 60 bone specimens from museums (We’re talking about 38,000-year-old bone); they repeated the sequencing for 35 times in the same clean room of extraction to avoid contamination with human DNA. Two years ago, the project of sequencing the Neanderthal genome started. They (Max Planck Institute & 454 Life sequencing) promised to end by this year. Well, they kept their promise. Frankly, some mitochondrial DNA sequences (mtDNA) have been published but contamination was the major defect in those published sequences. They collected more than 60 bone specimens from museums (We’re talking about 38,000-year-old bone); they repeated the sequencing for 35 times in the same clean room of extraction to avoid contamination with human DNA.

From the total 13 protein-encoding genes of the sequenced mtDNA, they identified only one with amino acids difference than the human sapien version. It is cytochrome c oxidase subunit 2 (COX2 – part of the respiratory chain), but even this difference has no significant effect on the functional domain of COX2. They hope to answer this questions in a few months: Why Neanderthals died out & human didn’t?!
We already know that Neanderthals & humans share 99.5% of the sequence, but answering questions about having a common ancestor & extinction through absorption (bred with humans) needs lots & lots of researches, collecting & sequencing samples at different time intervals to come with hypotheses. The mtDNA is not enough as Trinkaus (an expert on Neanderthal biology and human evolution) said: “The genome sequence data may tell us something about the selection of a couple of proteins, but it tells us nothing about language or social behavior.”
Image credits:
Reconstruction of a Neanderthal child from Gibraltar: http://en.wikipedia.org/
First complete Neanderthal genome sequenced: http://www.nature.com/
Tags: 454 pyrosequencing, genomics, human genome, human genome project, max planck institute, mtDNA, neanderthal
 2 Comments »
2 Comments »
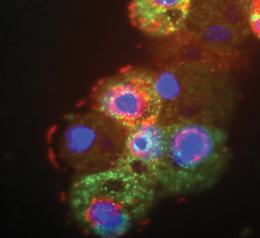 Once upon a time, in 2003, a French team discovered a giant virus infects amoeba. It was isolated from a cooling tower in the UK. They were so excited because it was so huge with a genome contains 900 protein-encoding genes (The words giant/ huge are totally hilarious. It’s not “Hulk”, it’s just a virus). It’s visible under the optical microscope. They named it Acanthamoeba polyphaga mimivirus (APMV). The prefix “mimi” is for mimicking microbe. Now, the same team “Raoult’s team” reported the isolation of another strain of those giant viruses but this time it was isolated from a cooling tower in Paris. They named it “mamavirus” because it was slightly larger than the previous giant virus (APMV), but it wasn’t alone. It was associated with its satellite, a small virus has 21 protein-encoding genes infects it, hijacks its viral factory making copies of itself, hindering the ability of the mamavirus to replicate/ make its own copies, so the number of the mamavirus drops in the infected amoebae. They named it Sputnik after the first man-made satellite. It’ll be the first isolated “Virophage”. How did I know about it? From the amazing blog of Dr. Ramy K. Aziz, “Microbes“. Once upon a time, in 2003, a French team discovered a giant virus infects amoeba. It was isolated from a cooling tower in the UK. They were so excited because it was so huge with a genome contains 900 protein-encoding genes (The words giant/ huge are totally hilarious. It’s not “Hulk”, it’s just a virus). It’s visible under the optical microscope. They named it Acanthamoeba polyphaga mimivirus (APMV). The prefix “mimi” is for mimicking microbe. Now, the same team “Raoult’s team” reported the isolation of another strain of those giant viruses but this time it was isolated from a cooling tower in Paris. They named it “mamavirus” because it was slightly larger than the previous giant virus (APMV), but it wasn’t alone. It was associated with its satellite, a small virus has 21 protein-encoding genes infects it, hijacks its viral factory making copies of itself, hindering the ability of the mamavirus to replicate/ make its own copies, so the number of the mamavirus drops in the infected amoebae. They named it Sputnik after the first man-made satellite. It’ll be the first isolated “Virophage”. How did I know about it? From the amazing blog of Dr. Ramy K. Aziz, “Microbes“.
The story won’t stop at this discovery. The discovery of the virophage will strongly suggest that “Viruses are alive” because they share something with other living domains of life, they can be infected, they can get sick, what makes all health-care providers totally thrilled because there’s something stronger than viruses which could be used to fight them, but “It’s too early to say we could use Sputnik as a weapon against big viruses or to modify them,” says co-author Bernard La Scola.
One more thing about Sputnik, 3 of its genes are closely related to APMV which suggests horizontal gene transfer between giant viruses caused by Sputnik. This is so “bacteriophagic”, reminds me with the whole insertion/ lysogenic mechanism between phages & bacteria. The isolated sequences from the ocean are closely related to the genome sequences of giant viruses & their satellite (Sputnik) . They infect plankton. “It suggests there are other representatives of this viral family out there in the environment,” Koonin says.
Image credits:
Giant mamavirus particles (red) and satellite viruses of mamavirus called Sputnik (green). http://www.nature.com/
Tags: APMV, horizontal gene transfer, mamavirus, sputnik, virophage
 No Comments »
No Comments »
To be frank, I was really underestimating these bacteria. Streptococcus iniae, bacteria bothering fish, what’s the big deal? But when I read that it cause them meningoencephalitis due to systemic dissemination resulting in death, it made me sad. They’re fish, you know, they don’t deserve such destiny. (We can’t say that about eating them, right?) Back to scientific details, S. iniae was isolated from Amazon dolphins (Inia) but it has a very wide host range, it can infect fresh & saltwater species like salmon, yellowtail & hybrid striped bass (HSB). It also can infect immunocompromised as well as elders from humans.
S. iniae are beta-hemolytic streptococci, they have M protein (present in cell membrane) then the capsular polysaccharide which interfere with phagocytosis. They also have phosphoglucomutase which make their cell wall rigid & resistant to peptide antimicrobials, besides their streptolysin S. I’ve to tell you this, this iniae is a walking disaster, just like the human version S. pyogenes.
So what scientists tell us this time? Thanks to 454 pyrosequencing & bioinformatics, they identified extra virulence factors of S. iniae. Regulations in S. iniae are done through a Mga-like Mgx loci (multiple gene regulator of group A streptococci). It regulates the virulence factors. It’s used to be known that M-protein is a component of that Mga. This time, discovery of extra components takes place, the M-like surface protein (simA) & C5a peptidase (scpI). A word about scpI, its role is to inactivate C5a (the complement component) to hinder the complement reaction & also has a role in adhesion to epithelial cells.
Actually they’re looking forward to using the mutant delta-simA as a live attenuated vaccine against S. iniae. (Vaccine for fish?) They made mutations in both C5a-like peptidase & simA, apparently simA had the leading role in the virulence of the studied strain of S. iniae. So a new approach of vaccination will be developed instead of the old M protein vaccination strategy which requires multimeric vaccination (to provide protection against several serotypes) & showes autoimmune response. Unlike the ordinary M protein vaccination, vaccination with the mutant delta-simA will result in development of humoral as well as cell-mediated immunity.
Image credits:
Fish – Cartoon: http://www.robdoyle.co.uk/
Tags: 454 pyrosequencing, fish, HSB, hybrid striped bass, live attenuated, S. iniae, vaccinology
 3 Comments »
3 Comments »
It’s hard for me, as a patriot, to read an article in EID about Plasmodium falciparum in ancient Egypt & shut up. The first emergence of Malaria in literature was when Hippocrates described the typical undulated fever which is highly characteristic for Malaria infection. Scientists made studies to identify P. falciparum‘s DNA in remains of a Roman infant (5th century) and another study on remains from 100-400 years ago.
They made their studies on 4000 years old mummies from Abydos, the Middle Kingdom tomb in Thebes West. Also samples from two different tombs from the New Kingdom until the Late Kingdom were taken. How did they know that those mummies had malarial infection? They simply searched for mummies had osteopathic evidence for chronic anemia. They ran PCR, a special technique called heminested PCR, using 18S rDNA primers. 2 of the 91 tested samples had chloroquine-resistance transporter (pfcrt) gene. I don’t know how, there wasn’t even chloroquine back there. They also ran immunological test which came positive for the P. falciparum histidine-rich protein-2 antigen.
Another thing came to my mind about Malaria. We must celebrate the launch of MalHaploFreq, the computer program which is designed for estimation of malaria haplotype frequencies in blood samples. Two awful facts about Malaria: There’s always drug resistance & multiple infections (the person can be infected by more than one type each one is resistant to an anti-malarial agent). All I know that they used Algorithm; this is really bad because I can’t stand math, and molecular markers to trace the spread of the drug-resistance & to predict the frequency of mutations.
Tags: malaria, MalHaploFreq, Plasmodium falciparum
 7 Comments »
7 Comments »
|











 Entries (RSS)
Entries (RSS)