Archive for the “Meet a Scientist” Category
Metagenomics is a culture independent approach that has contributed extensively to the study and understanding previously unidentified microbial communities. Seeking a further understanding of employing metagenomics in the study of the Red Sea microbial communities, we are pleased to interview Dr. Rania Siam, an Associate Professor in the Biology Department, the Director of the Biotechnology Graduate Program at the American University in Cairo (AUC) and an Investigator in the Red Sea Marine Metagenomics Project that is currently running at the AUC in collaboration with King Abdullah University of Science and Technology (KAUST), Woods Hole Oceanographic Institution and Virginia Bioinformatics Institute at Virginia Tech. Dr. Siam holds a Ph.D. in Microbiology and Immunology from McGill University. In addition, she held several post-doctoral positions at McGill Oncology Group, Royal Victoria Hospital, The Salk Institute for Biological Studies and The Scripps Research Institute. Since 2008, Dr. Hamza El Dorry (PI) and Dr. Siam (Co-PI) have been leading the Red Sea Metagenomics research team. The team is exploring novel bacterial communities in the Red Sea through actively participating in Red Sea expeditions for sampling and performing extensive molecular biology, genomics and computational analysis of the data.

1- Dr. Siam, thank you very much for accepting our request. Would you please explain for us the driving motives behind doing metagenomics research in the Red Sea?
The Red Sea is a unique environment in the region that remains to be explored. Thus, working on the Red Sea gives us the opportunity to perform essential research for the region. Furthermore, our main interest are the Red Sea brine pools that are unique environments in terms of high temperature, high salinity, high metal contents and low oxygen. Microbial communities living in these environments are known as extremophiles. The survival of extremophiles in such drastic conditions indicates their possession of genes with novel properties that underlie these unique survival characteristics. Thus, we are highly motivated to explore these novel microbial communities and their unique properties. In addition, we are interested in extracting biotechnological products from the Red Sea that can be beneficial as antimicrobials and anticancer agents.
2- Would you please outline for us the objectives of the project and the main activities inside and outside the laboratory?
In our project, one of our main aims is establishing a Red Sea marine genomic database to be accessed by scientists’ worldwide. Additionally, we are screening this database for biotechnological pharmaceutical products as enzymes and anticancer agents. There are three main activities in the project: sampling, molecular biology/genomics work and computational analysis of the data. Concerning sampling, it is a challenging process that requires rigorous planning where samples should be subjected to proper processing and storage till arrival to the labs. In labs, samples are subjected to different procedures starting from DNA extraction followed by Whole Genome Sequencing (WGS) to identify unknown genes or unique ones and help us understand novel microbial communities. This requires rigorous computational analysis to make sense of our data. Furthermore, we construct fosmid libraries for isolation and purification of genes of biotechnological interest as lipases and cellulases. In addition, we carry out 16s rRNA phylogenetic analysis on the microbial communities present in the samples.
3- Since the idea is novel, we would like to know about the nature of samples, the parameters and the challenges imposed during the process of sampling.
Basically, the sampling process requires well-equipped research vessels as the Woods Hole Oceanographic Institute ‘Oceanus’ and The Hellenic Center for Marine Research (HCMR) ‘Aegeo’. In addition, it is essential to have a team of physical oceanographics for adequate sampling.

http://www1.aucegypt.edu/publications/auctoday/AUCTodayFall09/Cure.htm
Image Source: AUC Today
We started with two different brine pools: Atlantis II Deep and Discovery Deep. As brine pools, these two regions are characterized by the presence of extremely harsh conditions as I mentioned before. In the 2008 and the 2010 KAUST Expedition to the Red Sea, we collected two forms of samples: Large volume water and sediments. In both cases, we face challenges during sample collection. Collecting large volume water samples can take up to 4 hours. In case of bad weather, it is nearly impossible to collect samples. Regarding the sediments, the heavy weight of the sediment core is the main challenge since it may drag people to the water during the sampling process.
The water samples are collected using CTDs (an acronym for Conductivity, Temperature and Depth), it is formed of 10 liter bottles that collect samples and measure the conductivity, temperature and depth, in addition to other parameters. CTDs are capable of measuring these physical properties from each meter of water. Accordingly, they are able to retrieve 2200 readings for each parameter at 2200 m depth. This is very beneficial as it allows us to correlate the physical and chemical parameters with the nature of microbial communities obtained from each sample.
4- What are the reasons behind the choice of Atlantis II Deep and Discovery Deep brine pools for study?
These sites are unique. Atlantis II Deep is 2200 meters below the water surface. It is characterized by having high temperature (68 °C), high heavy metal content, high salinity and little oxygen. Thus, these extremely drastic conditions are motivating us to explore the extremophilic microbial communities in this pool. Discovery Deep is adjacent to Atlantis II but the conditions are less harsh and varies in its heavy metal content. This encourages us to undergo comparative genomic analysis between the two regions.
5- Did the relatively new metagenomic approach of sequencing multiple genomes, combined with, the novelty of employing this approach in the study of microbial milieu in the Red Sea imply some unprecedented practical challenges in retrieving entire and authentic DNA sequences?
Yes, many challenges are present. For example, in case of sediments, many cells die. This in turn can make the process of DNA extraction more difficult. However, we managed to cope with this problem by quickly extracting the DNA from the sediments following its arrival to the labs and avoid freezing and thawing. Another problem with sediments is the presence of impurities that are co-extracted with DNA and interfere with the analysis. Concerning water samples, the main challenge is that the amount of DNA in the samples is very minute. This was dealt with by filtering large volumes of water up to 500 liters per sample.
6- Is recognizing and validating sets of data that are pointing out to specific patterns of microbial diversity or novel genes considered to be challenging?
Actually, we have retrieved enormous amount of data and a large percentage of the data has no match to sequences present in the other genomic databases. This has inspired us to think of new approaches for data analysis to identify the role of these novel sequences and seek collaborations with computational biologists.
7- Finally, do you think there are other environments in Egypt with unique properties which make it promising for employing metagenomics to discover novel genes and bacterial strains?
Yes, actually Egypt is very rich in environments with unique properties. For example, we have the deserts, Siwa‘s hot springs and the Nile. Many unique environments are yet to be explored and lots of research needs to be performed. Our natural resources are limitless.
Tags: 16s rRNA phylogenetic analysis, Aegeo, Atlantis II, AUC, Brine pools, Computational analysis, Computational biologists, CTDs, Culture-independent, Discovery Deep, DNA extraction, Drastic conditions, Expeditions, Extremophiles, Fosmid libraries, genomics, KAUST, Metagenomics, microbial communities, Molecular biology, Oceanus, Red Sea, Virginia Bioinformatics Institute, Virginia Tech., WGS, Woods Hole Oceanographic Institution
 2 Comments »
2 Comments »
This post on Microbe World entitled: “Software for Programming Microbes” did attract my attention, and I followed its source in the MIT Technology Review: An amazing article presenting a fascinating newly emerging technology of programming bacteria to do whatever we want them to do, produce drugs more efficiently, clean up oil spills, anything… useful! But the methodology was quite beyond my imagination.
I was extremely lucky to be able to interview Dr. Christopher Voigt, Associate Professor at the University of California, San Francisco, who is the project leader. Dr. Voigt has published over 34 articles indexed in Pubmed and you can find more about his projects on his lab website.
Now I will leave you with the interview! I thank Radwa for reviewing it.
 Dr. Christopher Voigt 1. May you please simplify the term “genetic circuit” to the micro-readers? What drove you to use software to genetically modify bacteria?
A genetic circuit functions like an electronic circuit, but uses biochemical interactions to do the computation. I am a computer programmer at heart and find living cells to be the ultimate challenge.
2. We used to hear a lot about the use of genetically modified bacteria in cleaning up toxicants or oil spills, producing drugs and biofuel. How is “programming bacteria” different from the “regular” definition of genetic engineering, which might be based on inserting a gene, a regulatory gene, or an operon that encodes for a certain needed functionality?
Genetic programming controls the timing and conditions under which those processes occur. It doesn’t refer to the pathways by which molecules are made or degraded.
3. In MIT Technology Review, you mentioned that like for a computer, programming bacteria is about writing a program to be encoded on a piece of DNA to implement a function. How can bacterial cells understand the code? How can the software make them sense the outer media?
The DNA contains codes for when molecules like proteins and mRNA should start and stop being produced and under what conditions. A protein can change its state when it senses a condition and bind to DNA to cause genes to be turned on or off. This acts like a sensor.
4. Honestly, I can’t imagine writing a piece of code to link bacterium to one another, because node->pRight!=NULL ? I just can’t imagine it. Is there any risk of overloading natural functions by accident?
No.
5. How can programing bacteria make use of quorum sensing?
Quorum signaling enables cells to be programmed to communicate with each other.
6. How can drug discovery and production benefit from programmable bacteria in the near future?
It makes it easier to access and control those pathways.
Tags: Christopher Voigt, genetic circuit, programmable bacteria, quorum sensing
 No Comments »
No Comments »
Dr. Betsey Dexter Dyer is a professor of Biology at Wheaton College, Massachusetts, USA. She received her Ph.D.  from Boston University. Among her research interests are symbiosis, evolution of cells, field microbiology and genomics. She is also part of the Genomics Research Group, a student project that she launched in collaboration with Mark LeBlanc, professor of Computer Science. Dr. Dyer has written several books, including from Boston University. Among her research interests are symbiosis, evolution of cells, field microbiology and genomics. She is also part of the Genomics Research Group, a student project that she launched in collaboration with Mark LeBlanc, professor of Computer Science. Dr. Dyer has written several books, including
The Modern Scholar: Unseen Diversity: The World of Bacteria (Audiobook), The Origin of Eukaryotic Cells (Benchmark Papers in Systematic and Evolutionary Biology, 9) (1986), Tracing the History of Eukaryotic Cells (1994 – with Robert A. Obar), Explore the World Using Protozoa (1997 – coauthor), A Field Guide to Bacteria (2003) and Perl for Exploring DNA (2007 – with Mark D. LeBlanc).
I recently read about her book “A Field Guide to Bacteria” and wanted to know more about her view on “Bacteriocentrism,” and how one becomes “bacteriocentric.” I was so lucky to be introduced to Dr. Dyer, who was kind enough to accept to be interviewed for Micro Writers, and immediately answered my questions about her book and her student project, Genomics Research Group, via email. So, here I am, sharing with you this very interesting interview.
1. Why did you decide to write the book “A Field Guide to Bacteria”? Field microbiology and symbiosis are among your research interests ; is this one of the reasons that made you dig deeper in bacterial populations and “think like a microbe”?
I first got the idea of a field guide when I was a graduate student and was very fortunate to be on a field microbiology expedition (in Baja California Mexico) with some world famous field microbiologists. I realized at once that a field guide should be written and one of them should write it and probably would! It did not occur to me that I would write it. It took me years to get to the point of being secure enough with my career. First I had to get a PhD, then a job, and then tenure. I also got married and had two children. Finally, about 15 years after that original idea, I realized that I had been accumulating enough information that I should begin to write. And so I did. However, I am still a bit surprised that nobody else wrote it.
I am naturally drawn to tiny things. I got a microscope for a present when I was 11 years old and it transformed some of my views of biology. I found that I loved the microscopic world. But I also like miniatures in general such as tiny furniture and dishes and things in dolls houses. I have in my library at home, some shelves devoted to a doll house and two miniature rooms.
2. What is meant by “becoming bacteriocentric”? And how does this lead to better understanding of the biology of bacteria?
We humans are mostly visual and auditory, the primary senses by which we perceive and analyze the world. It is probably impossible for us to be otherwise. Furthermore, we are gigantic and multicellular and terrestrial in marked contrast to the vast majority of organsims on Earth. Nonetheless, I think it is an excellent exercise for any biologist at least to try bacteriocentricity. The bacterial or microbial world is primarily olfactory and tactile. They are single celled (intimate with their environments), tiny and aquatic. I cannot avoid being anthropocentric but I can at least be more aware of the limitations of my size, habitat, and senses. My goal is to have as much humility as I can manage when I observe the world of microbes.
3. “Many groups of bacteria can be easily identified in the field (or in the refrigerator) without a microscope” and “Bacteria can be seen and smelled”, as a pharmacy student, I want ask how could that be achieved?
Well, do you have the book yet? There are many examples but the basis of all of them is that bacteria, when they are in an appropriate environment, are likely to do quite well: reproducing abundantly, taking in and transforming molecules, sending out wastes. In many cases (surprisingly many) the abundance is on a level perceptible by humans. The field marks just need to be revealed and interpreted. Otherwise, they may be easily overlooked or misunderstood. My first experience with this as a graduate student was being shown the distinctive pigmentations and odors of bacteria in sulfur cycles in Baja California.
4. You have a project with Dr. Mark LeBlanc, professor of Computer Science, called “Wheaton College Genomics Group.” Why did you do such a project for undergraduate students? How did it raise their potential?
One day about ten years ago, Mark asked me if I had any large datasets that his computer science students might analyze in their course on algorithms. It happens that I teach genetics and am fascinated with genomes. At that time, genome sequences were becoming more available at NCBI. I had not realized that it would be so easy to collaborate with a computer scientist. I had lots of questions about genomes and we just started right in with devising some answers. Right now, we are interested in characterizing horizontal transfer events of distantly related bacteria and archaea. There are hundreds of complete microbial genomes at NCBI and most have not been completely analyzed. Therefore, there is plenty for us and our students to do.
We ended up writing a book on the topic because we wee in need of a text that could be used both by biologists and computer scientists.
Tags: A Field Guide to Bacteria, bacteriocentric, bacteriocentrism, Betsey D. Dyer, biologists, computer scientists, field microbiology, Mark LeBlanc, NCBI, Perl, Perl for Exploring DNA, sulfur cycles, symbiosis, Wheaton College Genomics Group
 1 Comment »
1 Comment »
Not too long ago, I read about a research done at the Kennedy Institute of Rheumatology Division, which has identified a new ligand for Toll-like receptor 4. This receptor was previously known for activating the immune system through the detection of threats as lipopolysaccharide or gram-negative bacteria. The new ligand, Tenascin-C, is an extracellular glycoprotein, whose elevated expression in cases of inflammation provoked scientists to study its role in the process. 
The study noted that its presence was critical to maintain the ongoing inflammation seen in cases of rheumatoid arthritis. In reference to this study, the author stated “We have uncovered one way that the immune system may be triggered to attack the joints in patients with rheumatoid arthritis. We hope our new findings can be used to develop new therapies that interfere with tenascin-C activation of the immune system and that these will reduce the painful inflammation that is a hallmark of this condition”
I was able to contact Dr Kim Midwood and obtained this brief interview:
1. Do you have any speculations as to why Tenascin-C is overly expressed in certain individuals causing prolonged inflammation cases, whilst remaining within normal levels in others?
What regulates tissue levels of tenascin-C is not currently known and this is something that we are working on finding out.
2. From the different ligands of TLR4, why was Tenascin-C of particular interest in your research?
I have a long standing interest in how cell behavior is influenced by the extracellular environment, and in particular the role of extracellular matrix proteins in regulating cell phenotype during the response to tissue injury. For the last 10 years, I’ve been studying the role of tenascin-C – a protein specifically and transiently expressed upon tissue injury, but persistently expressed in chronic inflammatory diseases such as rheumatoid arthritis. This pattern of expression, plus the high homology of tenascin-C domains to other known pro-inflammatory matrix molecules or ‘DAMPs’ prompted us to investigate whether tenascin-C was an endogenous activator of the immune response and whether its persistent expression in RA contributed to disease pathogenesis.
3. What do you think the extent of similarity will be between the mice & human response to the Tenascin-C blockage?
I cannot predict how differently the mouse and human will behave.
4. Do you suspect a certain mechanism of the increase in inflammatory molecules caused by Tenascin-C?
We know that tenascin-C activates TLR4, activation of this receptor is well known to induced the expression of pro-inflammatory genes via activation of many intracellular signaling pathways.
5. How do you see the potential of such study for rheumatoid arthritis patients?
We plan to identify ways to inhibit the pro-inflammatory action of tenascin-C in the hope that this may be useful in reducing chronic inflammation in the joint.
Original research paper: Tenascin-C is an endogenous activator of Toll-like receptor 4 that is essential for maintaining inflammation in arthritic joint disease. Nature Medicine 15, 774 – 780 (2009). PMID: 19561617 (Vote for the abstract on Biowizard)
Image Credit: Davidson College Undergraduate Course
Tags: chronic inflammation, DAMPs, endogenous, inflammation, Interview, intracellular signal, Kim Midwood, ligand, pro-inflammatory, rheumatoid arthritis, rheumatoid therapy, Tenascin, TLR4, Toll-like receptor
 No Comments »
No Comments »
Meeting a scientist is like reading many books in few minutes, books of science and books of life. To me, though, meeting Dr. Niyaz Ahmed (1) was like reading a library. He is one of the most active advocates for Open Access (OA) and open evaluation of science. His support for OA is not just by words, but through sharing effectively in such purpose as he is a section editor of Microbiology and Genomics in PLoS ONE and a chief editor in Gut pathogen, the official journal of ISOGEM (the International Society for Genomic and Evolutionary Microbiology), in Sassari, Italy, of which Dr. Ahmed is a co-founder and the General Secretary. ISOGEM members work on developing post-genomic ideas to serve the public health and the environment. Dr. Ahmed is one of the faculty members of Faculty of 1000 Biology, the expert guide to the most important advances in biology.
Dr. N. Ahmed’s early start was in India as he graduated in Veterinary Medicine and obtained his Masters degree in Animal Biotechnology, then his PhD in Molecular Medicine. Currently, he is also based in India, in Hyderabad University as an Associate Professor of Biotechnology and a staff scientist in the Center of DNA Fingerprinting and Diagnostics in Hyderabad. This is not everything about him, his homepage reveals more, especially those pioneer achievements in the research world in India, like being a co-principal investigator of the Mycobacterium W genome program, India’s first whole genome sequencing project, and the most amazing part of his research is working on Helicobacter pylori chronological evolution and phylogeographic analysis.
Having read about all these achievements, I had many questions in mind, but, knowing how busy he is, I tried to reduce them to the following few questions that cover different areas of his activities. Dr. Ahmed generously agreed to answer them all.
Dr. Niyaz Ahmed, any microbiologist would be honored to meet you, and so am I. I am one of the “Micro Writers” blog members; we are young enthusiastic students from Faculty of Pharmacy, Cairo University in Egypt, mostly undergraduate and some graduate students, who are looking from the blog window towards the research world. Most of us wished someday to become scientists; “Micro Writers” is like the first real step on the right way. Our blog focuses on different areas of microbiology. It is like a message by students so far from Egypt, but hopefully from other places as well, to students all over the world. I read a lot about your achievements, especially those concerning the H. pylori phylogeographic analysis. As you have a great experience in research, reviewing articles and especially because you own a blog, I would like to ask you a few questions, and get them published with your answers on the blog, in order to teach and guide not only us, but the all students from everywhere.
Since you are working on the population structure of H. pylori, and on their chronological evolution, can the phylogeographic analysis of bacterial strains lead to infection control or innovative treatment? If yes, how?
-
N.A: It may not directly lead to product development as such, but, it can definitely explain how the spread-patterns of this organism are? and that has a lot of bearing on infection control in terms of tracking the infection sources. Also, knowing separate lineages in greater details offers accurate diagnostic development which in turn helps keep a tab on their emergence or otherwise.
If phylogeographic analysis can teach us about migration of some strains from one place to another, can we use it in a reverse way? I mean can we use this analysis to learn about the origin of mankind, and his migration routes?
-
N.A.: Indeed, we can do that. For a bacterial parasite to become a surrogate marker of human history (including migrations), it needs that its population structure mimics with that of the host. It is now established that H. pylori has most probably coevolved with its human host and thus its population structure is juxtapose to the human population structure. Therefore, we can use it as a marker of human history and geography.
Concerning the unpublished ideas of your studies, which suggest that bacterial proteins may be able to perform both the immune stimulatory and immune evasion tasks, the simple question is, do you think bacteria are smart? Do they have that smart controlling machinery that is able to distinguish between all these factors?
-
N.A.: The term ‘smart’ is reserved for humans. However, we can say that bacteria are highly adaptable. Several bacterial traits such as quorum sensing, community interactions (such as that seen in biofilm formation), molecular mimicry, immune evasion, dormancy etc. definitely point towards their being ‘intelligent’.
You talked about the host-pathogen relationship. I would like to add one more suggestion, which is that bacteria may face the immune system or the host body as a community of workers, not as independent cells. It is like the idea of sociomicrobiology in biofilms. Does this social behavior apply to other communities of bacteria other than bioflims, e.g. the community of H. pylori cells against the human defense system in the stomach?
-
N.A.: Indeed there is a body of evidence coming in via both in vitro and in vivo observations that H. pylori cells tend to form biofilms which is favored by gastric mucin and by mutations of luxS, and the cagE type IV secretion gene. This observation is important to understand H. pylori’s resistance to host immune responses and antibiotics, and in microenvironmental pH homeostasis which conditions and streamlines the growth and survival of H. pylori in vivo.
You mentioned at your homepage that strains of H. pylori in the Southern Asia are not life-threatening in the same time they may cause gastric cancer in the West. Do you think the lifestyle in Southern Asia, especially the food (extra-spicy food), might lead to evolution of these non-lethal strains, and vice versa in case of the western lifestyle?
Adoption of open access (OA) in science, isn’t it a huge responsibility, with great challenges, most probably the funding challenge? I mean, who will pay always for open access science?
-
N.A.: Open Access is a reality and the most practical approach to augment knowledge sharing towards science education, research and the practice of medicine and agriculture. Developing countries that have brains but no library budgets and that have been until now deprived of access to knowledge will be at a great benefit with this OA revolution. I do not see any challenge here as funding agencies such as NIH, Howard-Hughes Institute and Welcome Trust have already started subsidizing costs for OA in a big headway.
You started in India; did you feel some bias (in the international journal publishing) towards some western countries (e.g., USA, Germany, France, UK, etc.)? Do you think that the researchers from the developing countries should be encouraged in a way or another, especially against the fees and restrictions of publishing?
-
N.A.: I firmly believe that originality and novelty have no barriers. If your ideas are novel and your findings well defended, no one can stop them from seeing light of the day. Developing country scientists are equally respected – I can cite my own example, I was made Section Editor at PLoS ONE to oversee an extremely important area, microbial genomics; for this appointment, my affiliation and my geographical co-ordinates were not the important criteria but my standing in the field of medical microbiology, the efficiency with which I handled the issues and my professional commitment to the cause and ethos of OA publishing. However, there may be exceptions of which I have no idea. Nevertheless, this is a free world and if one has zeal and dedication, success is not that far. These days, publication restrictions based on novelty or being on ‘cutting edge’ or otherwise, are no issues. Developing country scientists are thus free to bring forward their research even on topics which may not be interesting to a few ‘glamour magazines’ of science, but they are welcome at descent venues such as PLoS ONE; they are free to even send in aspects of their folklore medicine, their local environmental problems, cropping, local biodiversity, energy production, their cultural and human anthropological aspects (and topics cutting across these) wherein no one is their competitor! Fees for publication is not a problem, PLoS and Biomed Central both waive off publication charges quite generously in almost all cases of genuine inability to pay.
In an old interview, you said that blogs are a good chance for young scientists to express their ideas for free, what about the older scientists in the developing countries who need more space to express their research activities, in the same time so many restrictions are imposed on them to get their papers published in the international journals?
-
N.A.: Old, and young, all need to be part of the dialogue. Even for busy academics, blogs are extremely helpful although they are no alternative to a proper publication of results or viewpoints. Blogs can effectively blend zeal and enthusiasm of ‘young Turks’ with the experience and patient advice of the ‘old guards’. Academic institutions in the developing countries should support such portals wherein students and faculty discuss research to arrive on novel interpretations and provocative ideas in an interdisciplinary environment of learning and enabling.
As an editor and reviewer in so many journals, what is your advice to some young Egyptian researchers like us in the “Micro Writers” blog who work with the fewest capabilities and with minimal resources, to get our papers published in an easier way?
-
N.A.: I love the Micro Writers Blog. You are the torch-bearers for all other undergraduate students in our countries of the developing world. My advice is try hard to deliver your best. Do not write on complaints – write about solutions! Do not present as Egyptian, or African, think globally, be a part of global community in the best interests of humanity, the environment and wellbeing of all creatures. I can quote from the Holy Qur’an: “Wallatheena Jaahadu Feena Lanahdiyannahum subulana” (The more you dedicate yourself and strive hard, the more will be the opportunities and paths widened for you from the Almighty). (2)
Finally, I would like to thank you Dr. Niyaz for the time you gifted for us to answer my questions with plentiful ideas, objects, and words. I am quite sure, each one who reads this interview will get benefited more than once.

Dr Niyaz Ahmed (left) co-ordinating a ‘student-scientist-administration’ meeting with Nobel Laureate Prof. Peter C. Doherty (center) and Australian High Commissioner to India, Mr John McCarthy (right).
———
(1): Dr Niyaz Ahmed, PhD
General Secretary, ISOGEM
Section Editor, PLoS ONE
Chief Editor, Gut Pathogens
Assoc. Editor, Ann Clin Microb Antimicrobials
Assoc. Editor, Acta Veterinaria Scandinavica
Faculty Member, Faculty of 1000 Biology
Corres. Fellow, Eur Helicobacter Study Group
(2): EL-Qur’an El-Karim, chapter #29 (El-Ankaboot), verse #69.
Image credits: Dr. Niyaz Ahmed.
Tags: Academic institutions in the developing countries, bacterial dormancy, biofilm, Biomed Central, cagE type IV secretion gene, chronological evolution, developing countries, Egyptian researchers, Faculty of 1000 biology, gastric cancer, gastric mucin, Gut pathogen, Helicobacter pylori, host-pathogen relationship, Howard-Hughes Institute, Hyderabad University, immune evasion, ISOGEM, luxS, microenvironmental pH homeostasis, NIH, Niyaz Ahmed, open access, phylogeographic analysis, PLoS ONE, quorum sensing, smart bacteria, the Center of DNA Fingerprinting and Diagnostics, Wellcome trust
 3 Comments »
3 Comments »
 Recently, I have been able to get in touch with Professor Jan Roelof van der Meer after reading about his work in EurekAlert in the field of color-coded bacteria. Currently, he is an associate professor at the Department of Fundamental Microbiology, University of Lausanne. Recently, I have been able to get in touch with Professor Jan Roelof van der Meer after reading about his work in EurekAlert in the field of color-coded bacteria. Currently, he is an associate professor at the Department of Fundamental Microbiology, University of Lausanne.
In a review published in collaboration with Professor Robin Tecon, the researchers explained why the bacteria were specifically useful in the detection & tracing back the age of oil spills, chemicals and other pollutants leaking into seawater and the soil. Since the bacteria are easily manipulated, researchers were able to genetically produce MBS “Microbe-Based Sensors” which produce specific reporter proteins when in contact with a certain pollutant. Such reporter proteins can then be detected merely by observation or instrumentally.
Enclosed within the review, this figure illustrates the concept of a bacterial sensor-reporter cell where the benzene-ring-look-a-likes represent the pollutants.
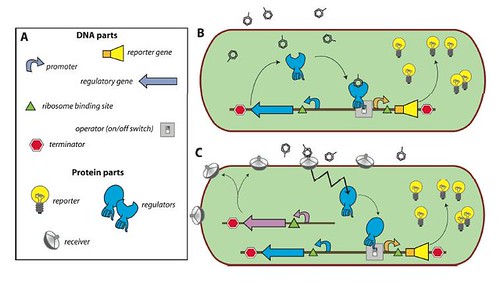
And I leave you with the interview:
1. Is there hope that MBS won’t just play a role in detection, but in cleaning up as well?
Normally not. To enhance biodegradation rates in the environment, one usually tries to stimulate the bacteria which are already present at the site. There are no cases where genetically modified bacteria were applied to clean up contamination.
2. How can this method trace back the age of a spill?
Interestingly, we found that there seems to be a specific pattern of dissolution of different compounds from oil. A fresh spill will first ‘show’ linear alkanes and compounds like benzene, toluene, ethylbenzene. Only later will polycyclic aromatic hydrocarbons, like naphthalene, appear. We had not seen this before, because one typically cannot measure the first phase of an oil spill, since this is detected only after a while.
3. Which method do you prefer in the genetic engineering of these MBS?
Depends. For E. coli, we use very classical cloning techniques involving plasmids. For other bacteria, we have to use transposon delivery methods mostly.
4. Is the acquired trait of producing a reporter protein passed on to future generations of the bacteria?
Normally yes. If the reporter construct is integrated in the genome of the bacteria, it is relatively stably maintained even without selection pressure for the marker. When the construct is on a plasmid, like in E. coli, one has to constantly keep the ‘pressure’ for the marker on the plasmid, usually an antibiotic resistance marker.
5. This field, as you kindly mentioned, started 20 years ago; what hope lies for its progess in the future?
My major hope is that people (industries, labs) finally apply the methods in their analysis as alternatives for costly chemical analysis. Further progress has to come from miniaturization, multiple target detections and improved methods to preserve the bacterial cells.
Tags: bacteria, biosensor, color coding, genetic engineering, Lausanne, MBS, oil spill, reporter protein, van der Meer
 2 Comments »
2 Comments »
|











 Entries (RSS)
Entries (RSS)